Inside a battery, there is a component that is thinner than a human hair, but plays an important role of prompting electrical reactions during charging and discharging the battery. What would that be? It is the current collector. Today, we’ll delve into the current collector.

What is current collector?
During the electrode manufacturing, a part of the battery manufacturing, slurry, a mixture of a variety of materials, is coated onto a current collector evenly. The current collector, a thin layer of about 10μm, induces electrochemical reactions during charging and discharging by delivering electrons from external circuits to active materials or vice versa.
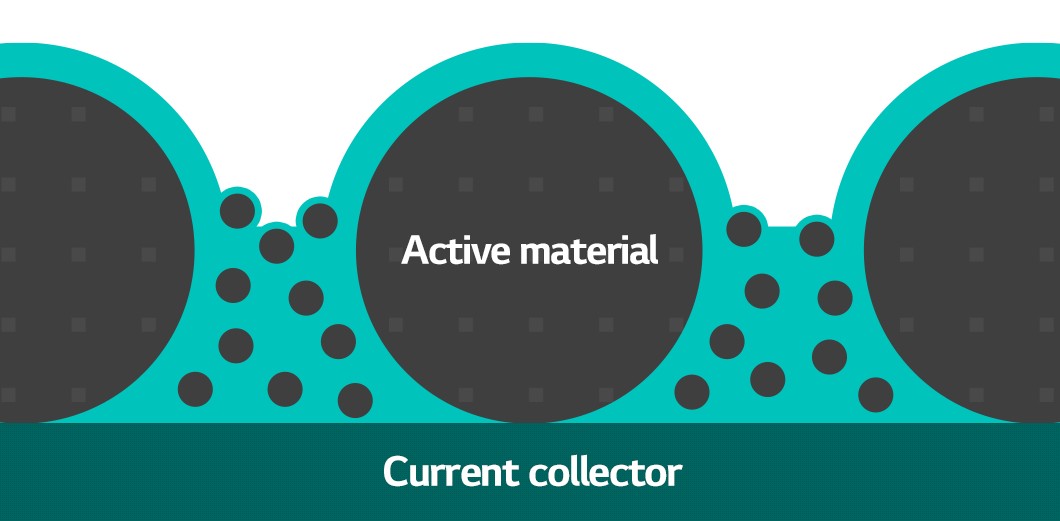
Ingredients of current collectors
Ingredients used for current collectors for lithium-ion batteries differ by electrode. The most commonly used material for the anode current collector is copper foil, since it offers high electrical conductivity and good stability against electrochemical reactions within the operating range of the carbon electrode. For the cathode current collector, aluminum foil is used, because it has a high electrical conductivity and stable electrochemical reactions at a high electric potential.

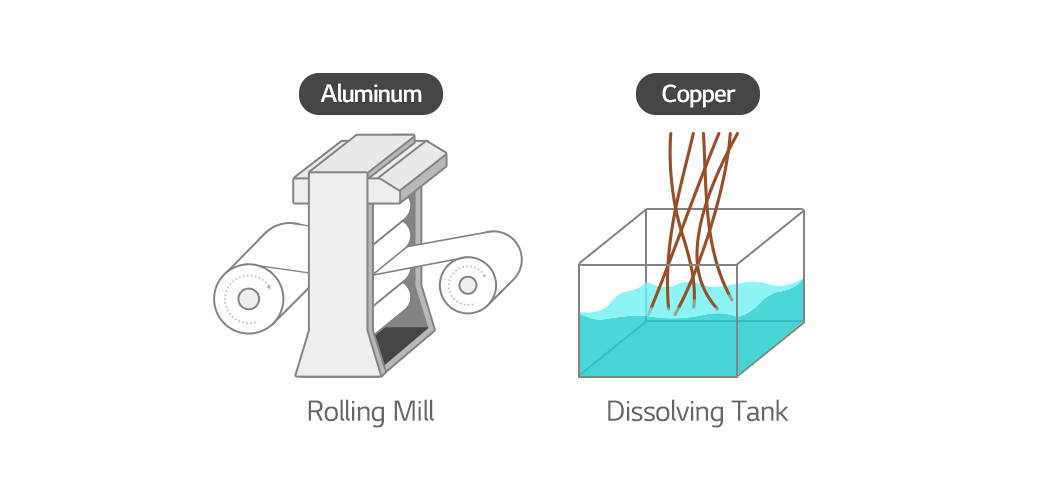
Manufacturing technique of current collectors
Each electrode is made through different methods. The aluminum foil for the cathode is obtained by rolling aluminum pieces thinly and evenly. And the copper foil for the anode is made by dissolving copper wire through electrodeposition.
The industry and academia are seeking to develop an anode current collector without materials that are relatively heavy and require much space in an attempt to further enhance the energy density. If developed, how will the new technology make a significant impact on the anode design for future batteries? You can look forward to fruitful achievements!





