
Battery performance can change significantly even with a single modification in materials. Ternary (NCM, NCA) and quaternary (NCMA) batteries are representative cathode chemistries that have evolved along this approach. Based on lithium cobalt oxide (LCO), these materials improve performance by combining nickel (Ni), manganese (Mn), and aluminum (Al).
As battery technology has advanced, demand for higher energy and greater capacity has continued to grow. To meet this need, the battery industry and researchers have focused on raising the nickel content to enhance energy density. This approach led to the emergence of high-nickel batteries. In this installment, we take a closer look at high-nickel batteries.
What is a High-Nickel Battery?
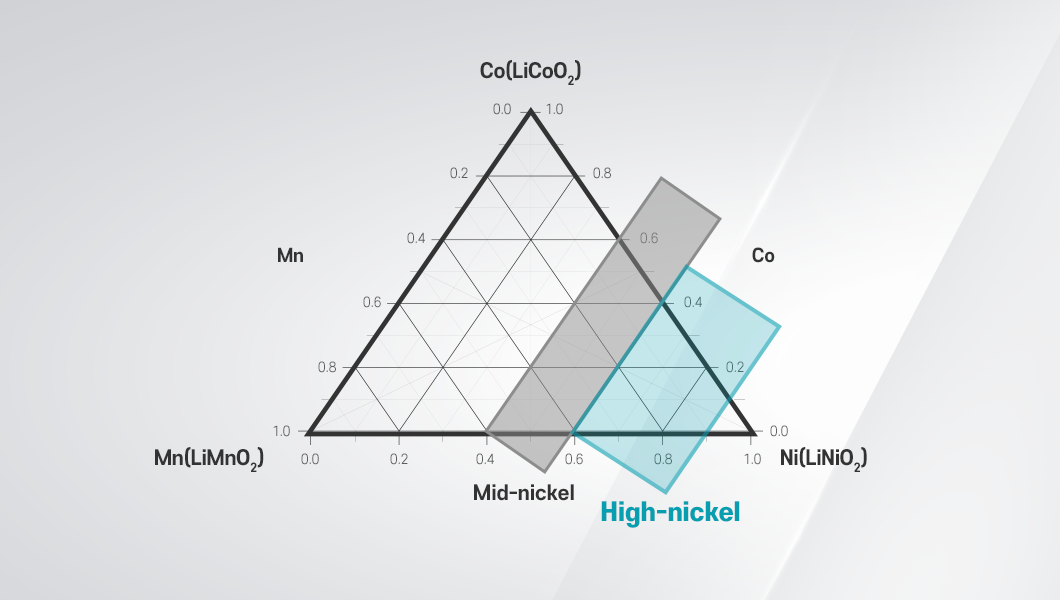
Among the elements that make up a cathode, nickel plays a critical role in determining how much energy a battery can store—that is, its energy density. For this reason, cathodes are classified by their nickel content. Batteries with a nickel content between approximately 40 to 60 percent are considered mid-nickel, while the proportions exceeding 60 percent are classified as high-nickel batteries. Recently, batteries with nickel content above 90 percent have also been under development.
As nickel content increases, reversible capacity during charge and discharge rises, enabling the battery to store more energy. This makes high-nickel batteries particularly well suited for electric vehicles, allowing longer driving range within the same volume.
What Led to the Rise of High-Nickel Batteries?
With the rapid growth of the electric vehicle market, customer demand for high-capacity and high-density batteries has intensified. The most direct way to address this demand was to increase nickel content in cathode materials to enhance energy density. While this straightforward principle guided the evolution of cathode materials, high-nickel batteries did not emerge from the outset.
Early lithium-ion batteries primarily used single-metal-based cathode materials such as lithium cobalt oxide (LCO), lithium nickel oxide (LNO), and lithium manganese oxide (LMO). However, each presented limitations in terms of capacity, safety, or lifespan. To overcome these limitations, ternary cathode materials (NCM, NCA) and quaternary cathode materials (NCMA) were developed by combining metals such as nickel, cobalt, manganese, and aluminum. These multi-metal systems provided the foundation for the evolution toward high-nickel batteries.
Ternary and quaternary cathode materials share a layered structure similar to lithium nickel oxide systems, ensuring structural stability. By adjusting the elemental ratios within this structure, performance and elemental balance could be optimized. As demand for higher capacity increased, these cathode materials evolved toward higher nickel content, leading to chemistries such as NCM712 and NCM811.
*View (Infographics #14) Structures of Cathode Materials
Technical Considerations for Commercializing High-Nickel Batteries
High-nickel cathodes offer structural advantages that enable high capacity. However, maintaining this high-capacity structure over the long term requires precise control of materials and structural design.
In high-voltage regions, lithium-ion batteries may undergo H2–H3 phase transitions, referring to significant structural changes during lithium insertion and extraction. Repeated H2–H3 phase transitions can lead to microcracks on particle surfaces or side reactions with the electrolyte.
Nickel and lithium also have similar ionic radii, which may cause cation mixing. In such cases, lithium diffusion pathways can be hindered, potentially affecting discharge rate or capacity characteristics. In addition, repeated charge–discharge cycles can alter the oxidation state of nickel, causing the particle surface to transition from a layered structure to a spinel or rock-salt structure. Over the long term, this can influence the battery’s thermal stability.

To manage these structural changes, various technologies are applied from the material design stage to enhance structural robustness. One such approach is bulk doping1, in which certain metal atoms within the lattice are substituted with dopants2 to strengthen metal–oxygen bonding. This helps mitigate cation mixing and high-voltage structural transitions, enhancing the intrinsic stability of the crystal structure. A doped lattice also supports more stable lithium diffusion pathways, reducing stress concentration and microcrack formation during charge–discharge cycling.
Another approach is surface coating. By suppressing side reactions and oxygen loss at the particle surface, coating slows structural changes and inhibits transitions to spinel or rock-salt phases. The coating layer reduces reactivity with the electrolyte, helps prevent microcracks, and supports more stable operation. As a result, lithium-ion transport becomes smoother, contributing to improved long-term cycle life.
The final approach involves precursor synthesis technology. By creating uniform particle size and morphology, it reduces localized stress during charge and discharge, mitigating microcracks and particle fracture.
LG Energy Solution’s High-Nickel Battery Strategy
LG Energy Solution continues to expand its portfolio to proactively address diverse customer needs. The company develops electric vehicle batteries across Premium, Standard, and Affordable segments, tailoring solutions to each category. Within the Premium segment, the strategy focuses on maximizing driving range, power output, and fast-charging performance. A key material supporting this strategy is high-nickel NCMA.
To further enhance high-nickel NCMA performance, LG Energy Solution is advancing single-crystal technology. While high-nickel NCMA was initially produced in the form of secondary particles, the company now applies single-crystal technology to achieve more precise control over material reactivity. Earlier development focused primarily on small-particle single crystals, but the scope has expanded to include medium-particle single crystals. This advancement increases packing density at the electrode level and enables higher cell energy density.
In addition, LG Energy Solution applies multi-doping, surface coating, and uniform coating process technologies to further improve the stability of high-nickel NCMA. These accumulated material and process capabilities provide the foundation for achieving higher nickel content and are expected to ultimately translate into greater energy density, longer driving range, and reduced total cost of ownership relative to driving range.

High-nickel batteries represent a key step toward achieving higher energy performance. LG Energy Solution will continue advancing high-nickel battery technologies to deliver higher-performance batteries with greater energy capacity. Through continuous innovation in battery materials, the company remains committed to strengthening both safety and performance in next-generation battery technologies.





