During a battery’s charge–discharge process, electric charge moves between the cathode and the anode, storing and releasing energy. However, some of this charge can be lost when side reactions such as electrolyte decomposition occur. Therefore, it is important to evaluate the ratio of the charge recovered during discharging to the charge supplied during charging. This ratio is a key indicator used to assess a battery’s efficiency and lifespan, known as Coulombic Efficiency (CE). Among various battery types, lithium-ion batteries exhibit relatively high Coulombic efficiency, which is why they are widely used across many applications. In today’s Battery Glossary, we take a closer look at the concept of Coulombic efficiency.
*Learn more about the structure and operating principles of lithium-ion batteries

What is Coulombic Efficiency?
Coulombic efficiency (CE) indicates how much electric charge a battery preserves without loss during a charge–discharge cycle. It is also referred to as Faradaic Efficiency or Current Efficiency.
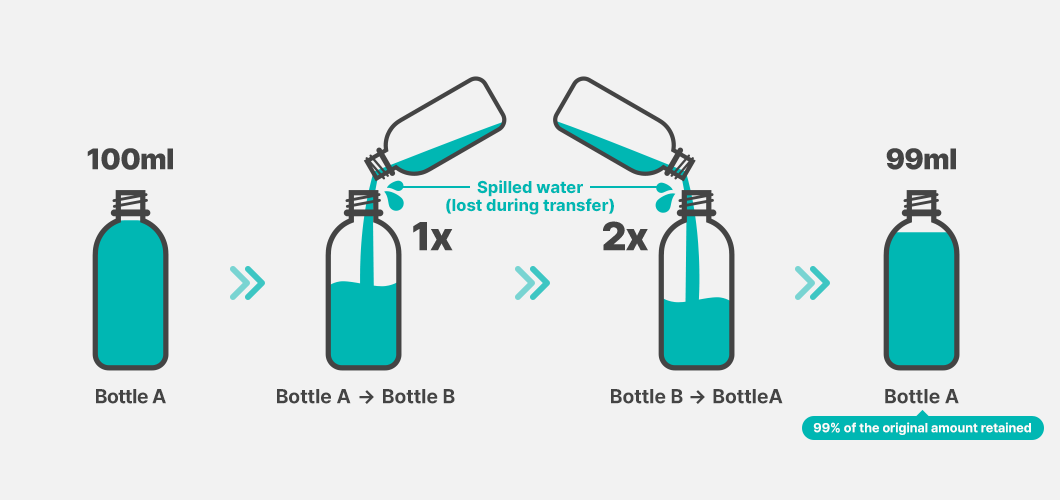
Imagine a bottle containing 100 mL of water. Suppose you pour all of this water into an empty bottle and then pour it back into the original bottle, repeating this process once. If a few drops are spilled along the way and only 99 mL remains, this means that 99% of the original water has been retained. This 99% ratio is analogous to Coulombic Efficiency.
In this analogy, the water represents the battery’s electric charge, while the act of transferring water between bottles represents the charging and discharging process (cycle) in which charge moves between the cathode and the anode. The spilled 1 mL corresponds to the amount of charge lost due to side reactions.
In other words, Coulombic Efficiency (CE) refers to the ratio of the charge (discharge capacity) that can be recovered during discharge to the charge (charge capacity) supplied during charging within a single charge–discharge cycle. It reflects a battery’s charge-retention performance, that is, how reversible the charge–discharge process can be.
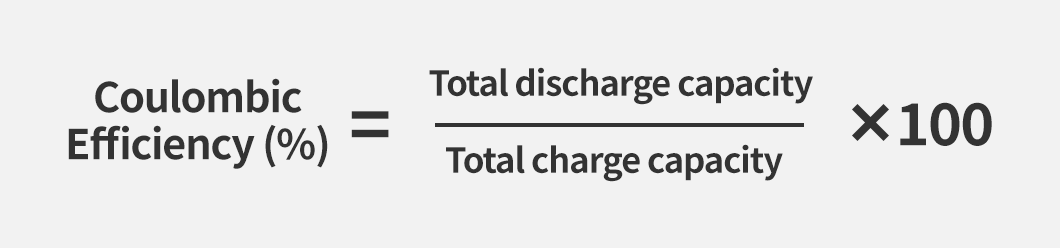
Coulombic Efficiency can be calculated using the formula below. It can be obtained by dividing the total discharge capacity (mAh or Ah) by the total charge capacity (mAh or Ah) and expressing the result as a percentage. For example, if a battery is charged with 100 mAh and discharged with 80 mAh, the Coulombic Efficiency is calculated as (80 ÷ 100) × 100 = 80%. This means that when 100 mAh of charge is supplied, only 80 mAh is actually usable.
If all of the charge supplied during charging were fully recovered during discharge within a single cycle, the Coulombic efficiency would be 100% (CE = 1). This would theoretically indicate a battery that operates permanently, with no side reactions occurring inside the cell. In reality, however, the charge recovered during discharge is always lower than the charge supplied during charging. This is because unavoidable electrochemical reactions, such as lithium loss, electrolyte decomposition, interfacial reactions, and changes in electrode structure, occur during battery storage and use.
As a result, repeated use of a battery, meaning repeated charge–discharge cycles, leads to a gradual decrease in efficiency and a reduction in battery lifespan. For this reason, Coulombic efficiency serves as a key indicator for estimating a battery’s cycle life and its performance limits.
As a related term, Initial Coulombic Efficiency (ICE) refers to the Coulombic efficiency measured during the first charge–discharge cycle of a battery. It represents the amount of charge lost due to side reactions during the initial activation process. A higher ICE indicates better initial efficiency and is advantageous for reducing long-term performance degradation.
In contrast, Energy Efficiency differs from Coulombic efficiency in that it also accounts for internal resistance and voltage losses and represents the ratio of actually usable energy. While Coulombic efficiency considers only charge, energy efficiency evaluates the ratio of usable output energy to input energy, measured in watt-hours (Wh). Understanding both indicators together allows for a more comprehensive assessment of a battery’s charge-retention capability and its actual energy utilization performance.
Why Coulombic Efficiency Matters in Batteries
Coulombic efficiency is an important indicator because it can be used to predict and evaluate battery lifespan, as well as stability, energy efficiency, operating costs, and system management accuracy.
■ Predicting battery life: A high Coulombic efficiency indicates minimal charge loss due to side reactions during charging and discharging, suggesting a longer battery lifespan.
■ Stable operation: A high coulombic efficiency, resulting from fewer internal side reactions, indicates that the battery can operate stably.
■ Cost efficiency: Higher Coulombic efficiency leads to longer battery life and reduced maintenance and operating costs. Conversely, low Coulombic efficiency results in energy loss and shorter replacement cycles, increasing overall costs.
■ Accurate system management: When Coulombic efficiency is accurately measured in a Battery Management System (BMS), the system can more precisely predict and control the state of charge (SoC), reducing the risks of overcharging and over-discharging.
*What is SoC? View Battery Glossary – SoC
Constant-Current and Constant-Voltage Charge–Discharge Tests for Measuring Coulombic Efficiency

Coulombic efficiency can be calculated using data obtained from constant-current (CC) and constant-voltage (CV) charge–discharge tests. These tests measure voltage, current, time, and capacity (current × time, measured in mAh or Ah), allowing Coulombic efficiency to be quantitatively evaluated.
First, the constant-current (CC) charge–discharge test maintains a constant current while charging and discharging the battery and is the most commonly used method for evaluating internal reaction characteristics and performance. The battery is charged by applying a constant current (I) until the voltage reaches a preset cut-off limit, at which point charging stops. The current direction is then reversed (−I) to discharge the battery. By repeating this process, the battery’s cycle characteristics and efficiency can be assessed.
In contrast, the constant-voltage (CV) charge–discharge test maintains a constant voltage throughout the charging process. When voltage is applied, the initial current is high but gradually decreases toward zero, indicating reduced internal charge transfer and stabilization within the battery. After a rest period, the battery is discharged by reversing the current direction.
*What are CC and CV? View Battery Glossary – CC/CV
We have taken a closer look at Coulombic efficiency, a key indicator used to evaluate battery lifespan and performance. By understanding the concept of Coulombic efficiency, it becomes easier to see how battery life gradually declines over time, as well as to better grasp the underlying operating principles and related battery concepts. In the next Battery Glossary, we will continue to introduce more fundamental and insightful battery concepts and terms.
Reversible: A property describing a process or state that can return to its original condition after a change.





